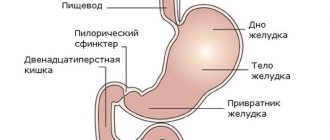
Lactase deficiency – a disease of the digestive system in children under one year old, it occurs quite often. Similar in symptoms to cow protein allergy, lactase deficiency is a slightly different disease.
Lactase is a special enzyme produced in the intestines of every person; its job is to break down lactose or milk sugar into smaller components (enzymes) for their further entry into the bloodstream.
Interesting fact : ancient people, namely people who lived more than 8,000 years ago (before the Ice Age) did not have the enzyme lactase in their bodies. This made it difficult for the species to develop and survive, since people were originally mammals. When “sapiens” developed the lactase enzyme in their intestines 7,500 years ago, milk became more digestible for them, children began to receive more nutrients, the survival rate of the species increased, and subsequently man evolved into what we are now.
From the name of the disease it can be understood that it is the insufficient activity of the lactase enzyme that causes the symptoms of lactase deficiency.
For various reasons, a child’s body may not produce lactase at all, or it may produce it partially; everything here is very individual and depends on the developmental characteristics of each child.
What is lactase deficiency?
Lactose and lactase - what is the difference?
What is lactase deficiency?
Complete lactase deficiency is a condition when a newborn’s body does not process lactose at all. Sometimes galactosemia is also included here, although galactosemia is a more complex disease due to its causes and symptoms.
Partial lactase deficiency is when the child’s body still processes lactose to some extent. In this case, the symptoms of the disease may be mild or absent altogether.
Lactase deficiency can also be primary or secondary. Let's explain in detail.
Briefly about lactose intolerance
Lactose intolerance is caused by insufficient digestion of lactose, a disaccharide component of milk and some dairy products. This carbohydrate is a biological source of energy, affects the development of the sense of taste, regulation of appetite, hormonal metabolism, as well as the processes of intestinal regeneration and peristalsis.
However, for this disaccharide to perform its functions, it must be digested in the small intestine. Deficiency of the intestinal enzyme (lactase), which is produced in the intestines and is responsible for digesting lactose (breaks it down into glucose and galactose), is insufficiently active or completely absent in people with intolerance. This becomes the reason that the body cannot cope with the processing of incoming dairy products.
The disease can manifest itself in various forms:
- congenital (alactasia) – the child’s body is not able to produce the enzyme lactase from the prenatal period. The first signs of the disease appear in the first days of a newborn’s life after receiving mother’s milk or an artificial formula containing lactose;
- primary (hypolactasia) - appears in adolescence or early adulthood, as a genetically determined lactase deficiency. The idea is that with age and an expanded diet, the body reduces the secretion of the enzyme;
- Secondary lactase deficiency in a child is considered acquired. It can appear, for example, as a result of taking antibiotics, non-steroidal anti-inflammatory drugs or against the background of chronic and infectious diseases of the gastrointestinal tract. Secondary lactase deficiency in a baby can be caused by diseases such as cystic fibrosis and Hirschsprung's disease.
Attention!
Temporary lactose intolerance can occur in premature babies because their intestines are not yet mature enough to produce lactase. In this case, the problem is temporary and disappears spontaneously when the child’s intestines begin to produce the necessary enzyme.
Symptoms of lactase deficiency
The body can signal that milk cannot be digested in different ways, depending on how much lactase it manages to create. The lower the amount, the more severe the symptoms. Milk indigestion can be observed 30 minutes and up to two hours after consuming a dairy product. Symptoms characteristic of lactase deficiency in a child:
- loose stools - lactose contained in milk is not able to overcome the intestinal wall to enter the bloodstream and, remaining in the intestines, retains water, which leads to diarrhea;
- stomach cramps - since lactose does not decompose, remains intact and absorbs large amounts of water, the accumulation of fluid stimulates intestinal motility, causing rhythmic contractions;
- excessive production of intestinal gases - undigested lactose, once in the large intestine, undergoes anaerobic fermentation and the production of organic substances, mainly lactic acid. They produce large amounts of gas, mainly carbon dioxide;
- bloating;
- nausea and vomiting;
- cramps, abdominal pain;
- rumbling in the stomach.
Symptoms usually disappear within a few hours.
You may be interested in:
Lactase deficiency in infants symptoms
Lactase deficiency in newborns
Lactose intolerance: symptoms
Intestinal obstruction in a child
Lactase deficiency
Attention!
Lactose intolerance manifests itself in different ways in children. Some people only experience discomfort when consuming large amounts of milk; for others, a small portion may cause symptoms. It also happens that a person does not suspect a problem until he includes a large amount of dairy products in his diet.
How to diagnose the disease
The diagnosis to confirm or exclude lactase deficiency in a child is made based on the observed symptoms that appear after consuming dairy products. Sometimes special diagnostic tests will be needed:
- lactose tolerance test - oral administration of the disaccharide and determination of glucose concentration. If the sugar level does not rise, lactose intolerance may be suspected;
- Hydrogen test – measures the amount of hydrogen exhaled. Such analysis, as a rule, is not prescribed for children;
- determination of stool pH - the acidity of the stool is examined. The presence of lactic acid may indicate a problem;
- genetic research.
Diagnosing congenital lactase deficiency in a newborn and infant is the most difficult. A biopsy of the small intestine is often required.
What else should be excluded from the diet?
It is necessary to reduce consumption to a minimum or completely eliminate:
- caffeine. Do not drink tea and coffee, they contain this substance;
- sugar;
- baking;
- do not drink alcohol in any form or strength;
- you need to carefully read the labels on products in stores, do not eat products with dyes and preservatives (this will be extremely difficult to do, since the main assortment of stores contains these substances);
- food with a high content of hot spices, pickles - cucumbers, mushrooms and others;
- no matter how bland it may be to eat dishes without seasoning - but this is necessary during breastfeeding;
- do not eat anything that can cause allergies in a child, for example, various exotic fruits or berries, and you should also not eat red vegetables;
- You should not eat yeast bread;
- legumes;
- grape.
Diet for lactose intolerance is very important.
How to treat the disease
It is difficult to talk about treatment in this case, as mentioned earlier, intolerance is a permanent condition that occurs due to the body’s inability to produce lactase. The only thing you can do is avoid milk and dairy products. It is recommended to completely stop taking:
- fresh milk and cream;
- mayonnaise;
- milk-based desserts: pancakes, chocolate, ice cream, cakes, cream-based creams, bird's milk, biscuits, cookies, etc.;
- products containing milk powder;
- any products that contain lactose.
Attention!
You need to know that lactose is used in the production of many foods. Individuals with severe intolerances should avoid anything that may contain even traces of this ingredient, such as chips, cereals, crackers, pies, margarine, some sauces, and sometimes even processed meat flavorings such as sausages, sausages or canned goods. Lactose may also be found in some medications (syrups, dragees)
In cases of congenital intolerance, a lifelong elimination diet is required. If lactose indigestion occurs in a later period (hypolactasia), then we are dealing with incomplete lactase deficiency. In this case, it is necessary to determine the degree of tolerance of the body and, accordingly, the amount of dairy products consumed. In case of secondary lactase deficiency in a child, temporary or periodic exclusion of milk is required.
Lactase deficiency: origin and ways of correction
The problem of lactase deficiency (LD) and lactose intolerance (as a clinical manifestation of LN) is as old as the world in the most literal sense of this expression. Lactose intolerance was first described by Hippocrates, approximately 400 BC, but understanding of the mechanisms of its development came only in the middle of the 20th century [1]. LN is associated with a decrease in the activity of the lactase enzyme of the brush border of enterocytes of the small intestine, which breaks down the milk sugar lactose. Traditionally, LI is divided into congenital, manifested from birth and caused by a mutation in the LCT gene on chromosome 21, primary (adult-type LI), associated with a genetically determined decrease in lactase activity after cessation of milk feeding, secondary, associated with damage to enterocytes in intestinal diseases, and transient LI of premature and immature children at the time of birth. The greatest importance, of course, is lactose intolerance in children in the first months of life, for whom human milk or infant formula are the only possible food products. At the same time, the most common and most attention-grabbing option is primary LN.
Humanity has been consuming milk and dairy products for thousands of years, which has led to the formation of a powerful dairy industry. In the world, cow's milk production dominates over the milk production of other ruminants, amounting to approximately 85%, followed by buffalo milk - about 11%, and goat and sheep milk - 2.4% and 1.4%, respectively. The share of cow's milk production is lower in the southern regions compared to the northern regions, and 80% of the world's goat's milk production is produced in the tropics. At the same time, in recent years, cow's milk production in developed countries has been decreasing, but increasing in developing countries. Particularly noteworthy is the large contribution to the production of cow's milk from India, a country in which a significant part of the population has adult-type LD (Table 1) [2, 3, 5].
The importance of milk consumption is primarily determined by the fact that milk is an important and accessible source of calcium. From the chart below, you can see that most of the calcium consumed is of animal origin. At the same time, in countries with low milk consumption, low calcium consumption is also observed. In this regard, correction of LI is a necessary factor in ensuring health, and when milk and dairy products are excluded from the diet of a child and an adult, it is necessary to compensate for the resulting calcium deficiency. Persons who limit the consumption of milk and dairy products for one reason or another should compensate for the resulting calcium deficiency from other sources, and it is hardly possible to solve this problem without prescribing medications (Fig.) [5].
There is also interesting data on the possible preventive effect of dairy products in relation to a number of cancers, in particular colon cancer, breast cancer, stomach cancer, as well as in relation to inflammatory bowel diseases, obesity and type 2 diabetes mellitus, however, this topic requires further in-depth study [6].
Lactose intolerance, a decrease in lactase activity after cessation of milk feeding, is a normal condition for all mammals, and only some representatives of Homo sapiens retained this ability throughout subsequent life.
Man first began to use ruminants in his economic activities in Mesopotamia between the 11th and 9th millennia BC. These were cows (Bos primigenius taurus), goats (Capra aegagrus hircus) and sheep (Ovis orientalis aries). At the same time, buffaloes (Bubalus bubalis) became domestic animals in India and China around 5000–7000 BC, and yaks (Bos grunniens) became domesticated animals in Tibet around 4500 BC. Domestic ruminants were already used in those years not only as a source of meat, but also milk and wool. Archaeological research has discovered the stomachs of these animals, in which milk was stored, which was processed into cheese under the influence of a residual amount of renin, a gastric enzyme. Cheese has been made from milk since at least 6500 BC. Sumerian cuneiform sources from the Third Dynasty of Ur (ca. 2000 BC) indicate the consumption of cheese, and cheese was produced in Egypt and Crete during the same period. In Ancient Rome, cheese was a common food product [7, 9]. Initially, the genotype of humans (like all mammals) included a decrease in lactase activity after the end of the breastfeeding period. Genetic studies have shown that mutations associated with lactose tolerance appeared around 10,000 BC, paralleling the domestication of ruminants [11]. The so-called "cultural-historical hypothesis" suggests that the continued high lactase activity of northern Europeans resulted from a selection process that allowed the population to rely on mammalian milk as an important component of the diet, especially in lean years. The opposite hypothesis defines a reverse cause-and-effect relationship, suggesting that widespread milk consumption led to the consolidation of lactose tolerance in the population, but this hypothesis seems less probable based on archaeological research [12–15].
In addition to being a source of energy and water, drinking milk provided a person with an effective source of calcium, providing higher bone density and preventing rickets [17]. This was more relevant for the northern regions (in particular, Northern Europe) in conditions of low insolation and a higher need for calcium with vitamin D deficiency. Accordingly, a higher frequency of lactose tolerance is observed in the northern regions of Europe, and intolerance - in the southern regions [5, 17, 19, 20]. Interestingly, lactose tolerance is associated with resistance to malaria, a phenomenon studied primarily in the Fulani people of Africa, but the mechanisms behind this relationship remain unknown [21].
The LCT gene, which encodes lactase and determines the breakdown and, therefore, tolerance of lactose, has a length of 49.3 kb, is located on the long arm (q) of chromosome 2 at position 21 and contains 17 exons (NCBI Reference Sequence NG_008104.1) [23] . For primary LN, a genetic polymorphism has been described affecting the replication factor MCM6, one of the highly conserved proteins of the MCM group required for the initiation of replication in eukaryotic cells (primarily the LCT-13910C.T polymorphism in intron 13 and LCT-22018G.A in intron 9 of the MCM6 gene ) [25–27]. The LCT-13910CT and LCT-13910TT genotypes are associated with lactose tolerance and, being present in one allele in a heterozygous state, have a dominant effect, ensuring the breakdown of lactose, while the LCT-13910CC genotype in the absence of the LCT-13910T allele leads to lactose malabsorption [26 , 27]. There is parallelism between the genotypes LCT-13910C.T and LCT-22018G.A. Individuals with the LCT-13910CC genotype have the LCT-22018GG variant, while those with LCT-13910CT often have LCT-22018GA, with LCT-13910TT associated with LCT-22018AA, with the exception of a small number of observations in Finland [27], China [28] and in Brazilians of Japanese descent [29]. These lactose malabsorption genotypes are widespread in Europe, while other lactose tolerance/intolerance alleles are present in other regions of the world [30].
In Europe, the prevalence of primary FN varies from 2% in Scandinavia to 70% in some regions of Italy [31]. Its prevalence in the white population of the United States is 20%; in Asia it is close to 100%. The prevalence of FN in adults in Sweden, Denmark is about 3%, Finland, Switzerland - 16%, England - 20-30%, France - 42%, Southeast Asian countries, among African Americans in the USA - 80-100%, European Russia - 16–18%. However, not all people with LI require correction.
Only those patients with LI who have clinical manifestations require treatment. Deviations from the norm in the results of diagnostic methods, which are not accompanied by corresponding symptoms, are also not grounds for therapy.
In case of congenital and primary LI, the patient’s treatment is based on reducing the amount of lactose in the diet, up to its complete elimination. In parallel with this, therapy is carried out aimed at correcting intestinal dysbiosis and other symptomatic treatment.
In case of secondary LN, the main attention should be paid to the treatment of the underlying disease, and reducing the amount of lactose in the diet is a temporary measure that is carried out until the mucous membrane of the small intestine is restored.
Reducing the amount of lactose in the diet can be achieved by eliminating lactose-containing products, primarily whole milk, from the diet.
This method is quite acceptable for adults and older children with adult-type LI and secondary LI. In this case, the consumption of fermented milk products, butter, and hard cheeses is often allowed. The possible reduction in calcium intake with a dairy-free diet should be considered.
In infancy, if the child is breastfed, the optimal approach is to use lactase preparations, which are mixed with expressed breast milk and break down lactose without affecting its other properties. Lactase producers for the production of corresponding drugs and dietary supplements (and in most cases, products containing lactase are registered as dietary supplements) are yeast, most often Kluyveromyces fragilis, Kluyveromyces lactis or molds Aspergillus niger and Aspergillus oryzae.
Children who are bottle-fed should be switched to full or partial nutrition with low-lactose (lactose-free) breast milk substitutes, choosing a diet with the maximum amount of lactose that does not cause the appearance of clinical symptoms and an increase in the content of carbohydrates in the feces.
The diet is supplemented by symptomatic therapy, which depends on the child’s condition and the severity of secondary disorders. If exicosis develops, therapy aimed at correcting the water and electrolyte balance is required. Also, the development of intestinal dysbiosis can determine the indications for the use of probiotics.
In the future, after symptoms have stopped in children with LI, it is necessary to monitor the carbohydrate content in feces over time and, when symptoms return, reduce the amount of lactose.
In breastfed children in the absence of lactase drugs, similar tactics are used.
Complementary feeding dishes (porridge, vegetable purees) for children of the first year of life with LI, naturally, should not contain lactose. The calendar for introducing complementary foods does not differ from that for children without disabilities.
In children over one year of age and adults with LI, products containing lactose are excluded from the diet (Table 2). It is advisable to replace milk and dairy products with specialized low-lactose dairy products: low-lactose milk, cream, sour cream, etc. in quantities determined by age-related needs. These patients usually tolerate fermented milk products (kefir, yogurt) well.
The problem of moderate lactase deficiency can be solved by consuming fermented milk products with low lactose content, due to the release of β-galactosidase into the gastrointestinal tract by live probiotic bacteria that can survive in the gastrointestinal tract [18]. Unique in this regard among other fermented milk products is yogurt, like buttermilk or sweet acidophilus. They are well tolerated by people with lactase deficiency and promote the breakdown of lactose thanks to the bacteria Lactobacillus bulgaricus and Streptococcus thermophilus [18, 24], which release β-galactosidase in the intestine. The administration of probiotics improves the enzymatic activity of intestinal microorganisms, increases the digestion of lactose, reduces the symptoms of lactase deficiency, slowing down the transit of contents through the digestive tract. Pasteurization of fermented milk products reduces the number of living bacteria [18, 24]. A clinical study of capsules containing Bifidobacterium longum and yoghurt fortified with Bifidobacterium animalis proved effective in relieving symptoms of lactose intolerance [10]. A similar study in children (ages 5 to 16 years) found that consuming milk with Lactobacillus acidophilus or yogurt containing Lactobacillus lactis and Streptococcus thermophilus reduced symptoms of lactase deficiency, compared with a group of children consuming unfortified milk [18]. It is known that by fermenting lactose with lactic acid microorganisms, it is possible to utilize up to 20% of lactose as much as possible (up to 4% of residual lactose), which is ineffective from the point of view of lactase deficiency. The possibility of producing low-lactose fermented milk products using the enzyme preparation β-galactosidase obtained from the yeast Kluveromyces lactis was studied. The rate of fermentation of milk hydrolyzed with β-galactosidase was studied, both before the introduction of the starter and simultaneously with its introduction. In one case, the starter was added to pre-hydrolyzed milk (the fermentation process was combined with the reserve process; in the other, the starter was added directly with the β-galactosidase enzyme (0.2%, concentration 2000 NEL/g). Reducing the ripening process and increasing the water-holding capacity in samples with the simultaneous addition of enzyme and starter can be explained by the fact that the products of enzymatic catalysis activate the starter microflora. The content of residual lactose in control samples (without enzyme) was 4.0–3.8%; in test samples with preliminary hydrolysis of lactose 1.2– 1.0%; in samples with simultaneous addition of enzyme and starter – 1.0–0.8% [34, 37].
It should be noted that lactose has become widely used in the food and pharmaceutical industries in recent decades. It is used as a sweetener and as a foaming agent, gives products a pleasant taste and forms an attractive texture in the production of baked goods, drinks (both non-alcoholic and alcoholic, in particular beer), meat products (for example, in the production of burgers), etc. Important that lactose is not fermented by mushrooms, and this avoids excess production of carbon dioxide and alcohols. Lactose can be present in processed meats (sausages, pates, etc.), margarine, bread, potato chips, sauces, processed cheeses, protein supplements, and beer. In the pharmaceutical industry, lactose is widely used as a filler in pharmaceuticals. In this regard, the volume of industrial production of lactose is growing year by year [5, 32, 33, 35]. Accordingly, the list of lactose-containing non-dairy products is also expanding.
The duration of therapy is determined by the nature of the disease. In case of congenital LI, a low-lactose diet is prescribed for life. With transient LI of prematurity, many children gradually regain the ability to tolerate lactose over several months (although this does not exclude the development of primary LI). Canceling the diet is carried out under individual monitoring of lactose tolerance.
With secondary hypolactasia, symptoms of FN are transient. Patients are treated for the underlying disease; Along with this, a low-lactose diet is prescribed in accordance with the principles outlined above. When the normal structure and function of the small intestinal mucosa are restored, lactase activity is restored. Therefore, when the underlying disease resolves (remission) after 1–3 months, the diet can be gradually expanded, although even here a strictly individual approach is required [36].
Taking into account the intestinal dysbiosis that develops in clinically manifest LI, it is advisable in this situation to prescribe probiotics. They can be prescribed as stand-alone medications, but some manufacturers include them in formulas for artificial feeding.
Maxilak® and Maxilak Baby do not contain casein (milk protein) and lactose (milk sugar), they are hypoallergenic and can be used even with high allergic sensitivity. Maxilak® and Maxilak® Baby contain bifidobacteria and lactobacilli of various strains and can be used to maintain endogenous flora in case of secondary lactase deficiency.
Maxilak® is available in two forms:
- Sachet Maxilac® Baby from 4 months of age (Lactobacillus acidophilus LA-14 - 1.11 × 108 CFU; Lactobacillus casei CBT (1) - 1.11 × 108 CFU; Bifidobacterium lactis BL-04 - 1.11 × 108 CFU; Lactobacillus paracasei Lpc-37 - 1.11 × 108 CFU, Lactobacillus plantarum Lp-115 - 1.11 × 108 CFU, Lactobacillus rhamnosus GG - 1.11 × 108 CFU, Lactobacillus salivarius Ls-33 - 1.11 × 108 CFU; Bifidobacterium bifidum BF-2 - 1.11 × 108 CFU; Bifidobacterium longum BG-7 - 1.11 × 108 CFU);
- • Maxilak®, capsules for children over 3 years of age and adults, including pregnant women (Lactobacillus helveticus - 9.00 × 108 CFU; Lactococcus lactis - 9.00 × 108 CFU; Bifidobacterium longum - 6.75 × 108 CFU; Bifidobacterium breve - 4.50 × 108 CFU; Streptococcus thermophilus - 4.50 × 108 CFU; Lactobacillus rhamnosus - 4.50 × 108 CFU; Lactobacillus casei - 2.25 × 108 CFU; Lactobacillus plantarum - 2.25 × 108 CFU; Bifidobacterium bifid um — 2.25 × 108 CFU).
In addition to the release form (capsules and sachets), Maxilak® and Maxilak® Baby differ from each other in the dosage of active ingredients. Thus, one Maxilac® capsule contains 4.5 billion CFU (colony-forming units of bacteria), and one Maxilac® Baby sachet (sachet) contains 1 billion CFU. All strains included in Maxilak® and Maxilak® Baby are of original origin and are included in the list of European quality, which proves a high level of safety. Studies of the acid resistance of Maxilak® probiotic strains at pH = 2 for two hours demonstrated high viability. C. Chapman et al. [4] proved that, unlike individual strains, multistrain probiotics are characterized by higher antimicrobial activity (p < 0.05). Unlike modified starch, pectin and other fibers, oligofructose selectively stimulates the growth and metabolic activity of certain types of bacteria (bifidobacteria and lactobacilli), without affecting the growth of other groups of bacteria (fusobacteria, bacteroides, etc.), and inhibits the growth of potentially pathogenic bacteria (Clostridium perfringens, Clostridium his/lyt, Enterococci groups), oligofructose also increases the protective effects of bifidobacteria. In the production of Maxilak®, a unique capsule protection technology MURE® (Multi Resistant Encapsulation) is used - the capsule is covered with a shell that protects its contents and preserves the viability of bacteria even when exposed to aggressive factors of the external and internal environment, therefore the bacteria present in Maxilak® are protected from the acidic contents gastric juice, bile salts and digestive enzymes. The theoretical premises underlying the developed synbiotic provide for the correction of disturbed intestinal microflora, which usually arise as a result of acute infections, the course of chronic diseases, the use of antibacterial drugs, imbalances in the functioning of organs and systems of the macroorganism, including lactase deficiency.
To summarize, we can conclude that primary LI is a widespread individual feature that in most cases does not require medical intervention. LN should be called a disease only in case of obvious clinical manifestations. Of course, the problem becomes most important in children in the first months of life, for whom milk is the main food product. As for older children and adults, it should be borne in mind that the absence of dairy products in the diet can lead to calcium deficiency in the body, and therefore drug correction of this condition is necessary.
Literature
- Beja-Pereira A., Luikart G., England PR et al. Gene-culture coevolution between cattle milk protein genes and human lactase genes // Nat Genet. 2003; 35: 311–313.
- Boll W., Wagner P., Mantei N. Structure of the chromosomal gene and cDNAs coding for lactase-phlorizin hydrolase in humans with adult-type hypolactasia or persistence of lactase // Am J Hum Genet. 1991; 48:889–890.
- Cavalli-Sforza LL Analytic review: some current problems of human population genetics // Am J Hum Genet. 1973; 25:82–104.
- Chapman TM, Plosker GL, Figgitt DP VSL#3 probiotic mixture: a review of its use in chronic inflammatory bowel diseases // Drugs. 2006; 66(10):1371–1287.
- Coelho M., Luiselli D., Bertorelle G. et al. Microsatellite variation and evolution of human lactase persistence // Hum Genet. 2005; 117:329–339.
- Enattah NS, Sahi T., Savilahti E., Terwilliger JD, Peltonen L., Jãverlã I. Identification of a variant associated with adult-type hypolactasia // Nat Genet. 2002; 30: 233–237.
- Evershed RP, Payne S, Sherratt AG, Copley MS, Coolidge J, Urem-Kotsu D, Kotsakis K, Ozdogan M, Ozdogan AE, Nieuwenhuyse O et al. Earliest date for milk use in the Near East and southeastern Europe linked to cattle herding // Nature. 2008, 455, 528–531.
- Gerosa J., Skoet J. Milk Availability: Trends in Production and Demand and Medium-Term Outlook. Available online: www.fao.org/docrep/015/an450 e/an450 e00.pdf (accessed on 26 May 2015). ESA Working paper no. 12–01 February 2012.
- Global Consumption of Dairy Products. Canadian Dairy Information Centre. Available online: https://www.dairyinfo.gc.ca/index_e.php?s1=dff-fcil&s2=cons&s3=consglo (accessed on May 26, 2015).
- He T, Priebe MG, Zhong Y, Huang C, Harmsen HJ et al. Effects of yogurt and bifidobacteria supplementation on the colonic microbiota in lactose-intolerant subjects // J Appl Microbiol. 2008, 104: 595–604.
- International Dairy Federation (IDF). World Dairy Situation 2014; Bulletin No. 476/2014; IDF: Brussels, Belgium, 2014.
- Kretchmer N. Lactose and lactase // Sci Am. 1972; 227:71–78.
- Leonardi M., Gerbault P., Thomas MG, Burger J. The evolution of lactase persistence in Europe. A synthesis of archaeological and genetic breastfeeding evidence // Int. Dairy J. 2012, 22, 88–97.
- Lokki AI, Järvelä I., Israelsson E., Maiga B., Troye-Blomberg M., Dolo A., Doumbo OK, Meri S., Holmberg V. Lactase persistence genotypes and malaria susceptibility in Fulani of Mali // Malar J. 2011, Jan 14; 10:9.
- Lomer MC, Parkes GC, Sanderson JD Review article: lactose intolerance in clinical practice - myths and realities // Aliment Pharmacol Ther. 2008, Jan 15; 27 (2): 93–103.
- Malmström H., Linderholm A., Lidén K., Storå J., Molnar P., Holmlund G., Jakobsson M., Götherström A. High frequency of lactose intolerance in a prehistoric hunter-gatherer population in northern Europe // BMC Evol . Biol. 2010, 10, 89, I.
- Mattar R., Monteiro MS, Silva JMK, Carrilho FJ LCT-22018 GA single nucleotide polymorphism is a better predictor of adult-type hypolactasia/lactase persistence in Japanese-Brazilians than LCT-13910 CT // Clinics (São Paulo). 2010; 65:1399–1400.
- Montes RG, Bayless TM, Saavedra JM, Perman JA Effect of milks inoculated with Lactobacillus acidophilus or a yogurt starter culture in lactose-maldigesting children // J Dairy Sci. 1995, 78: 1657–1664.
- Nei M., Saitou N. Genetic relationship of human populations and ethnic differences in reaction to drugs and food // Prog Clin Biol Res. 1986; 214:21–37.
- Ozdemir O., Mete E., Catal F., Ozol D. Food intolerances and eosinophilic esophagitis in childhood // Dig Dis Sci. 2009; 54:8–14.
- Ranciaro A., Campbell MC, Hirbo JB, Ko W.-Y., Froment A., Anagnostou P., Kotze MJ, Ibrahim M., Nyambo T., Omar SA et al. Genetic origins of lactase persistence and the spread of pastoralism in Africa // Am. J.Hum. Genet. 2014, 94, 496–510.
- Rasinperä H., Savilahti E., Enattah NS et al. A genetic test which can be used to diagnose adult-type hypolactasia in children // Gut. 2004; 53:1571–1576.
- Salque M., Bogucki PI, Pyzel J., Sobkowiak-Tabaka I., Grygiel R., Szmyt M., Evershed RP Earliest evidence for cheese making in the sixth millennium BC in northern Europe // Nature. 2013, 493, 522–525.
- Savaiano DA, Abou El Anouar A., Smith DE, Levitt MD Lactose malabsorption from yogurt, pasteurized yogurt, sweet acidophilus milk, and cultured milk in lactase-deficient individuals // Am J Clin Nutr. 1984, 40: 1219–1223.
- Sherratt A. The secondary exploitation of animals in the Old World // World Archaeol. 1983, 15, 90–104.
- Silanikove N., Leitner G., Merin U., Prosser CG Recent advances in exploiting goat's milk: Quality, safety and production aspects // Small Ruminant Res. 2010, 89, 110–124.
- Simoons FJ Primary adult lactose intolerance and the milking habit: a problem in biology and cultural interrelations: II. A cultural historical hypothesis // Am J Dig Dis. 1970; 15: 695–710.
- Szilagyi A. Adaptation to Lactose in Lactase Non-Persistent People: Effects on Intolerance and the Relationship between Dairy Food Consumption and Evaluation of Diseases // Nutrients. 2020 Aug 13; 7(8):6751–6779.
- Troelsen JT Adult-type hypolactasia and regulation of lactase expression // Biochim Biophys Acta. 2005; 1723: 19–32.
- US Food and Drug Administration - Problems Digesting Dairy Products? Available online: https://www.fda.gov/forconsumers/consumerupdates/ucm094550.htm#intolerance (accessed on May 26, 2015).
- Xu L., Sun H., Zhang X. et al. The –22018 A allele matches the lactase persistence phenotype in northern Chinese populations // Scand J Gastroenterol. 2010; 45: 168–174.
- Zecca L., Mesonero JE, Stutz A. et al. Intestinal lactase-phlorizin hydrolase (LPH): the two catalytic sites; the role of the pancreas in pro-LPH maturation // FEBS Lett. 1998; 435:225–228.
- Zecca L., Mesonero JE, Stutz A. et al. Intestinal lactase-phlorizin hydrolase (LPH): the two catalytic sites; the role of the pancreas in pro-LPH maturation // FEBS Lett. 1998; 435:225–228.
- Danilov M. B. Activity of β-galactosidase of microorganisms used in the production of dairy products // Storage and processing of agricultural raw materials. 2001. No. 7. pp. 30–31.
- Mngram CJ, Liebert A., Swallow DM Population Genetics of Lactase Persistence and Lactose Intolerance; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012.
- Mukhina Yu. G., Chubarova A. I., Geraskina V. P., Belmer S. V., Gasilina T. V., Borovik T. E., Roslavtseva E. A., Skvortsova V. A., Yatsyk G. V. Working protocol for the diagnosis and treatment of lactase deficiency in children // Issues. det. diets 2016; 14(1):64–69.
- Ripelius K., Dvinsky B. M. Maxilact - enzyme treatment of milk solves the problem of lactose intolerance // Dairy industry. 1995. No. 5. pp. 23–24.
S. V. Belmer, Doctor of Medical Sciences, Professor
Federal State Budgetary Educational Institution of Russian National Research University named after. N. I. Pirogova, Moscow
Contact Information
Lactase deficiency: origin and ways of correction / S. V. Belmer
For citation: Attending physician No. 2/2018; Page numbers in the issue: 41-46 Tags: infancy, feeding, lactose, intolerance
Buy an issue with this article in pdf